
cpt code for ultrasound guided biopsy of breast
Accurate CPT coding for ultrasound-guided breast biopsies, specifically CPT code 76645, is crucial for appropriate reimbursement and reflects the procedure’s complexity.
Overview of Breast Biopsy Procedures
Breast biopsy procedures are essential for diagnosing breast abnormalities, ranging from benign conditions like fibrocystic changes to malignant tumors. Ultrasound-guided breast biopsy utilizes real-time imaging to precisely target suspicious areas, ensuring accurate tissue sampling. These procedures encompass both core needle biopsies, extracting small tissue cylinders, and incisional biopsies, removing a larger portion of tissue through a small incision.
The process begins with patient positioning and ultrasound probe placement to visualize the target lesion. Sterile technique is paramount throughout. Following local anesthesia, a biopsy needle is guided by the ultrasound beam into the lesion. Multiple samples are often obtained to ensure representative tissue for pathological analysis. Post-procedure compression is applied to minimize hematoma formation.
The choice between core needle and incisional biopsy depends on lesion characteristics and clinical judgment. Accurate documentation of the procedure, including lesion location, needle gauge, and number of cores obtained, is vital for correct CPT coding, particularly CPT 76645.
Importance of Accurate CPT Coding
Precise CPT coding is paramount in healthcare billing, and ultrasound-guided breast biopsies are no exception. Utilizing the correct code, primarily CPT 76645, ensures appropriate reimbursement from payers like Medicare, which in 2010 averaged a global fee of $89.85. Incorrect coding can lead to claim denials, delayed payments, and potential compliance issues.
Accurate coding reflects the skill and resources involved in these procedures, including the radiologist’s expertise, ultrasound equipment utilization, and sterile supplies. Under-coding undervalues the service, while over-coding risks accusations of fraud. Detailed documentation supporting the chosen code is essential, outlining the procedure’s specifics.
Furthermore, staying abreast of CPT updates and changes, including Category III codes for new technologies like MRI-ultrasound fusion guidance (Koelis Imagio), is crucial. Proper coding directly impacts a facility’s financial health and ensures sustainable healthcare delivery for breast care patients.
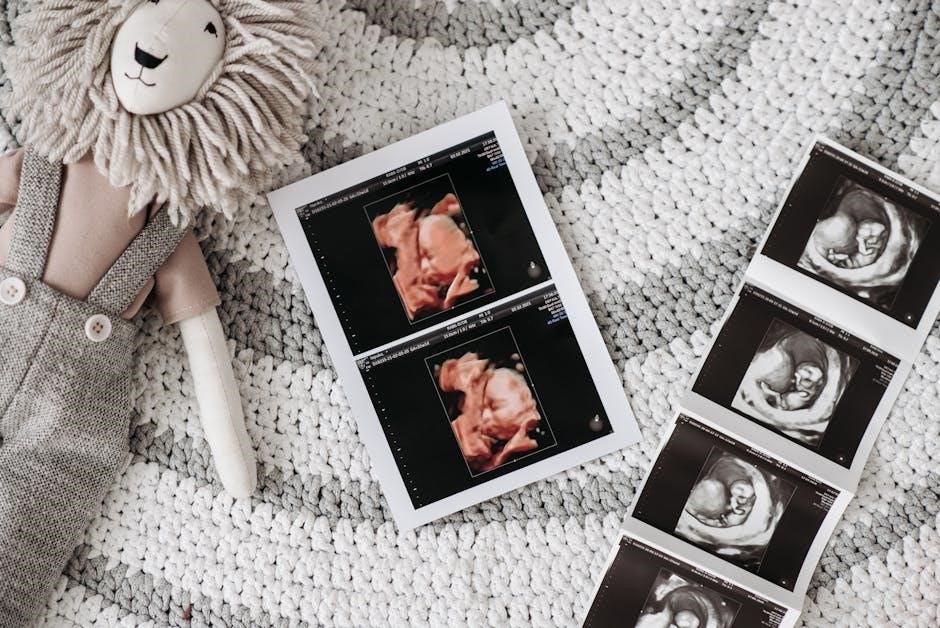
CPT Codes for Ultrasound-Guided Breast Biopsy
The primary CPT code for ultrasound-guided breast biopsy is 76645, encompassing imaging guidance for percutaneous needle placement or localization.
CPT Code 76645: Ultrasound Guidance for Breast Biopsy
CPT code 76645 specifically describes ultrasound guidance for the placement of a needle for breast biopsy procedures. This encompasses both core needle biopsies and incisional biopsies performed under real-time ultrasound visualization. The code isn’t simply for the ultrasound itself, but for the guidance it provides during the biopsy process, ensuring accurate targeting of suspicious lesions within the breast tissue.
It’s important to understand that 76645 represents the imaging component, and must be billed in conjunction with the appropriate biopsy code representing the tissue acquisition itself. The 2010 Medicare reimbursement averaged a global fee of $89.85, highlighting the need for accurate coding to maximize appropriate payment. This code is fundamental when documenting procedures like thyroid and breast ultrasound-guided interventions. Proper application of 76645 is vital for compliant billing and reflects the skill and precision involved in ultrasound-guided breast biopsies.
Detailed Breakdown of 76645 Components
CPT code 76645 isn’t a monolithic service; it comprises several key components essential for accurate billing. These include real-time ultrasound imaging to visualize the lesion, meticulous patient positioning for optimal access, sterile preparation of the skin, and precise needle guidance to the target area.
The radiologist or qualified healthcare professional must actively use the ultrasound throughout the entire biopsy procedure – not just for initial targeting. This continuous visualization ensures accurate sample acquisition and minimizes the risk of non-diagnostic results. Documentation should clearly reflect this real-time guidance. Furthermore, the code covers the interpretation of the ultrasound images during the procedure to adjust needle trajectory as needed.
Essentially, 76645 represents the cognitive work and technical skill involved in using ultrasound to safely and effectively obtain a breast tissue sample for pathological examination. It’s the imaging component that elevates the biopsy’s precision.
When to Use Modifier +59 with 76645
Modifier +59, indicating distinct procedural services, is crucial when multiple biopsies are performed during the same session under CPT code 76645. It’s applied when biopsies target separate lesions, even within the same breast, or when biopsies are performed in both breasts during a single encounter. Simply put, if the biopsies aren’t a continuation of the same procedure on the same lesion, +59 is likely needed.
Incorrectly reporting multiple biopsies with only 76645 can lead to claim denials or accusations of unbundling. Thorough documentation is paramount; each lesion biopsied must be clearly identified, along with its location and the rationale for the separate procedure.

The +59 modifier demonstrates to payers that each biopsy represents a unique service requiring independent skill and resources. Remember, the decision to use +59 hinges on the distinct nature of each biopsy performed, ensuring accurate representation of the services rendered and appropriate reimbursement.

Billing Considerations for Ultrasound-Guided Biopsies
Proper billing for CPT 76645 requires meticulous documentation, understanding payer guidelines, and awareness of Medicare reimbursement rates, which historically averaged $89.85.
Medicare Reimbursement Rates for CPT 76645
Medicare reimbursement for CPT code 76645, ultrasound guidance for breast biopsy, has historically been a point of discussion among providers. Data from 2010 indicates an average global fee of approximately $89.85, though current rates are subject to annual updates and geographic variations.
It’s essential to consult the latest Medicare Physician Fee Schedule (MPFS) to determine the precise reimbursement amount for a specific location and year. Reimbursement typically includes a professional component (physician’s fee) and a technical component (facility fee), which may be billed separately depending on the provider’s setting.
Factors influencing reimbursement can include the complexity of the case, the use of modifiers (like +59 for distinct procedural services), and adherence to proper documentation guidelines. Accurate coding and comprehensive documentation are vital to maximize appropriate reimbursement and avoid claim denials. Providers should regularly review Medicare updates and coding resources to stay informed about changes affecting CPT 76645 reimbursement.
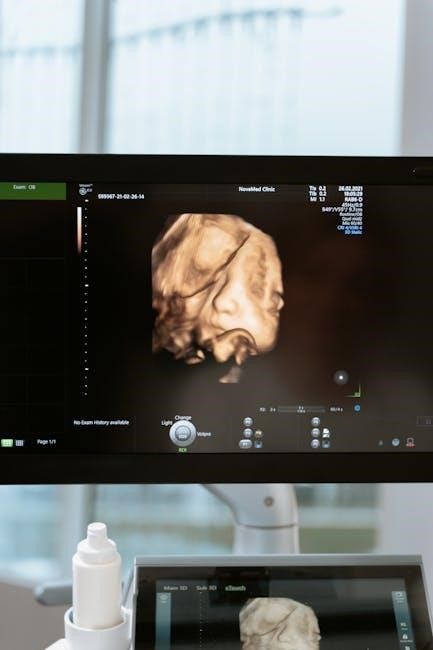
Coding for Incisional vs. Core Needle Biopsies
While CPT code 76645 covers ultrasound guidance for any breast biopsy, the subsequent coding for the biopsy itself differs based on the technique. An incisional biopsy performed via an open procedure, as described in examples, would be coded separately using surgical CPT codes – potentially 17284, 17274, or 17264, depending on the extent of tissue removed.
Core needle biopsies, however, don’t have a specific ultrasound-guided code beyond 76645. The pathology examination of the core biopsy specimen is reported using separate CPT codes within the pathology section (e.g., 88305). It’s crucial to accurately represent the type of biopsy performed.
Incorrectly coding an incisional biopsy as a simple needle biopsy, or vice versa, can lead to significant billing errors and potential audit issues. Thorough documentation detailing the biopsy method is paramount for accurate code assignment and appropriate reimbursement. Remember to always code the guidance (76645) and the biopsy procedure itself.
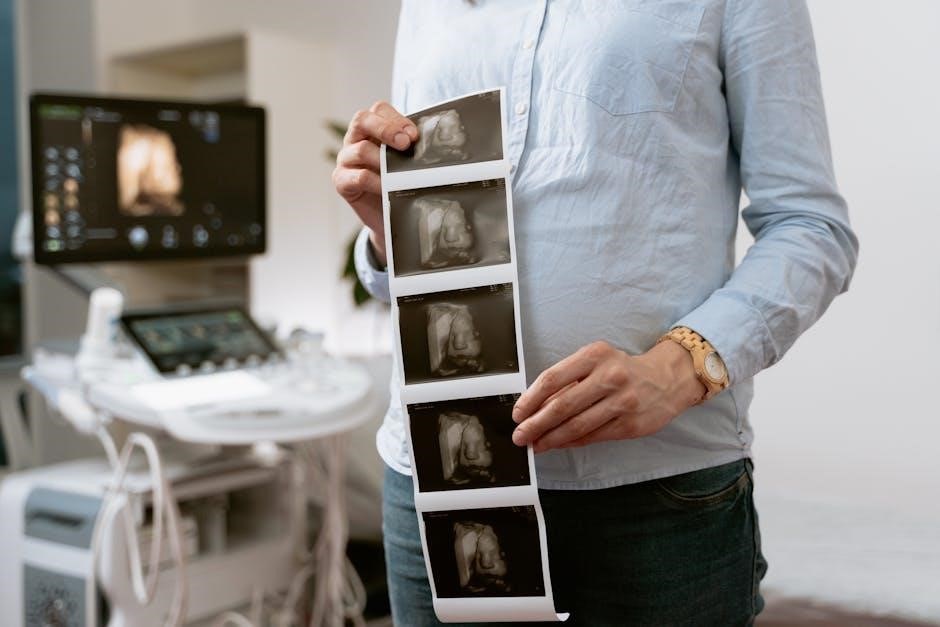
Documentation Requirements for Proper Coding
Comprehensive documentation is essential for justifying the use of CPT code 76645 and ensuring accurate billing for ultrasound-guided breast biopsies. The medical record must clearly demonstrate the medical necessity for the biopsy, including the suspicious findings identified on the initial ultrasound examination.
Detailed notes should outline the ultrasound guidance process itself – transducer placement, needle visualization, and real-time imaging during sample acquisition. Specify the biopsy technique used (core needle, incisional, etc.) and the number of samples obtained. Include any complications encountered and how they were addressed.
Furthermore, documentation must support any modifiers used, such as +59 when multiple biopsies are performed at separate locations. Pathology reports confirming the specimen type and findings are also crucial. Insufficient or ambiguous documentation can lead to claim denials or audits, emphasizing the importance of meticulous record-keeping for compliant coding practices.

Related CPT Codes & Procedures
Additional CPT codes relate to breast biopsies, including pathology examination codes and diagnostic ultrasound codes; MRI-ultrasound fusion guidance (CPT code for Imagio) is also relevant.
CPT Codes for Pathology Examination of Biopsy Specimens
Following an ultrasound-guided breast biopsy utilizing CPT code 76645, proper coding for the subsequent pathology examination is essential for complete billing. Several CPT codes cover these services, varying based on the specimen type and complexity of the examination. For example, codes in the 88300-88399 range are frequently used.
CPT 88300 represents a Level 1 microscopic examination, while higher-level codes (88304-88309) denote increasingly complex procedures, potentially including special stains or immunohistochemistry. The specific code selected depends on the pathologist’s work performed. Accurate documentation detailing the specimen source (core biopsy vs. incisional biopsy), the number of fragments, and any special studies conducted is paramount.
Furthermore, codes for ancillary studies, such as receptor analysis (e.g., estrogen and progesterone receptor testing), should be appended when applicable. Proper sequencing of codes is also vital; typically, the biopsy code (76645) is listed first, followed by the pathology examination code. Consistent and detailed documentation ensures accurate claim submissions and minimizes audit risk.

CPT Codes for Breast Ultrasound (Diagnostic)
While CPT 76645 specifically addresses ultrasound guidance for a breast biopsy, diagnostic breast ultrasounds performed prior to the biopsy are coded separately. The primary CPT code for a diagnostic breast ultrasound is 76645, however, additional codes may apply depending on the extent and complexity of the examination.
CPT 76645 covers a limited ultrasound examination, while more comprehensive studies, potentially including Doppler imaging or evaluation of multiple quadrants, may require additional codes. For instance, if the ultrasound is performed for screening purposes, different codes might be applicable based on payer guidelines.
Accurate documentation detailing the indication for the ultrasound (e.g., palpable lump, screening, follow-up), the areas examined, and any findings is crucial for appropriate code selection. It’s important to distinguish between diagnostic and screening ultrasounds, as reimbursement policies often differ. Proper coding ensures accurate billing and reflects the clinical services provided before the guided biopsy.
CPT Codes for MRI-Ultrasound Fusion Guidance (Koelis Imagio)
The use of MRI-ultrasound fusion guidance systems, like Koelis Imagio, for breast biopsies introduces specific CPT coding considerations beyond the standard 76645 for ultrasound guidance. As of January 7, 2026, the AMA issued a new Category III CPT code specifically for Imagio (OA/US) Breast. This code acknowledges the advanced technology and precision offered by fusion guidance.
Currently, the Category III code is often reported alongside 76645 to accurately reflect the complete procedure. However, Category III codes typically have limited payer coverage, so verifying specific insurance policies is essential. The fusion guidance enhances the accuracy of biopsy targeting, particularly for small or non-palpable lesions identified on MRI.
Proper documentation must clearly demonstrate the use of the fusion technology, including details of the MRI-ultrasound registration process and the system utilized (e.g., Koelis Imagio). This detailed documentation supports the medical necessity and appropriate billing for the advanced procedure.

Coding Updates and Changes
The AMA continually evaluates CPT codes; Category III codes, like those for MRI-ultrasound fusion, may evolve with technological advancements and increased utilization data.
AMA Category III CPT Codes for New Technologies
The American Medical Association (AMA) utilizes Category III CPT codes to track emerging medical technologies and procedures, including advancements in breast biopsy techniques. These codes, unlike Category I or II, are temporary and often represent innovative approaches not yet widely adopted or fully established.
For instance, the use of MRI-ultrasound fusion guidance systems, such as Koelis Imagio, initially fell under Category III coding. This allowed for data collection on utilization, safety, and efficacy before potential transition to more permanent Category I status. Currently, the AMA assesses new technologies impacting breast biopsy, potentially leading to the creation or modification of Category III codes.
Coders must stay informed about these evolving codes, as they directly impact billing and reimbursement. The availability of Category III codes facilitates appropriate payment for novel procedures while providing the AMA with valuable information for future CPT coding decisions. Regularly checking the AMA website and coding resources is essential for accurate reporting.
Impact of Chinese Guidelines on Coding Practices
While primarily focused on domestic healthcare, the 2020 Chinese guidelines for ultrasound and breast disease management offer insights relevant to global coding practices, particularly concerning breast biopsy procedures. These guidelines emphasize standardized diagnostic pathways, including ultrasound-guided biopsies, influencing procedural volumes and potentially, coding patterns.
The Chinese guidelines highlight the importance of accurate diagnosis of both benign and malignant breast conditions, driving the need for precise biopsy coding. Although direct application to US CPT coding is limited, the emphasis on thorough documentation and standardized reporting mirrors best practices universally.
Furthermore, the Chinese experience with lower reimbursement rates for CPT code 76645 (as noted in some reports) serves as a cautionary tale regarding payer policies and the potential impact on access to advanced diagnostic procedures. Understanding international perspectives, like those from China, can inform advocacy efforts for fair reimbursement and appropriate coding valuations within the US healthcare system.
Future Trends in Breast Biopsy Coding
The landscape of breast biopsy coding is poised for evolution, driven by technological advancements and a growing emphasis on precision medicine. The emergence of new technologies, like MRI-ultrasound fusion guidance systems (e.g., Koelis Imagio), is prompting the development of new CPT Category III codes to accurately reflect these procedures.
Expect increased scrutiny of documentation requirements to support CPT 76645 and related codes, ensuring appropriate utilization and preventing coding errors. Tele-ultrasound and remote guidance may also necessitate new coding paradigms as these modalities gain traction.
Furthermore, ongoing monitoring of payer policies, including Medicare reimbursement rates, will be critical. The potential for bundled payment models or value-based care initiatives could significantly impact how breast biopsies are coded and reimbursed. Staying abreast of AMA updates and industry best practices will be essential for accurate and compliant coding in the future.